Furan And Maleic Anhydride Reaction
| |||
 | |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name Furan-2,5-dione[2] | |||
| Other names Maleic anhydride[2] | |||
| Identifiers | |||
| CAS Number |
| ||
| 3D model (JSmol) |
| ||
| Beilstein Reference | 106909 | ||
| ChEBI |
| ||
| ChEMBL |
| ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.003.247 | ||
| EC Number |
| ||
| Gmelin Reference | 2728 | ||
| PubChem CID |
| ||
| RTECS number |
| ||
| UNII |
| ||
| Un number | 2215 | ||
| CompTox Dashboard (EPA) |
| ||
| InChI
| |||
| SMILES
| |||
| Properties | |||
| Chemic formula | C four H 2 O 3 | ||
| Molar mass | 98.057 g·mol−1 | ||
| Appearance | White crystals or needles[iii] | ||
| Odor | irritating, choking[3] | ||
| Density | 1.48 g/cmiii | ||
| Melting betoken | 52.8 °C (127.0 °F; 325.ix K) | ||
| Boiling point | 202 °C (396 °F; 475 K) | ||
| Solubility in h2o | Reacts | ||
| Vapor pressure | 0.2 mmHg (twenty°C)[3] | ||
| Magnetic susceptibility (χ) | -35.8·10−6 cm3/mol | ||
| Hazards | |||
| GHS labelling: | |||
| Pictograms |    | ||
| Signal word | Danger | ||
| Hazard statements | H302, H314, H317, H334, H372 | ||
| Precautionary statements | P260, P261, P264, P270, P272, P280, P285, P301+P312, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P304+P341, P305+P351+P338, P310, P314, P321, P330, P333+P313, P342+P311, P363, P405, P501 | ||
| NFPA 704 (fire diamond) | three one ane | ||
| Flash point | 102 °C (216 °F; 375 K) | ||
| Explosive limits | 1.four%-7.ane%[3] | ||
| Lethal dose or concentration (LD, LC): | |||
| LDl (median dose) | 465 mg/kg (oral, mouse) 850 mg/kg (oral, rat) 875 mg/kg (oral, rabbit) 390 mg/kg (oral, guinea pig) 400 mg/kg (oral, rat)[4] | ||
| NIOSH (US wellness exposure limits): | |||
| PEL (Permissible) | TWA 1 mg/miii (0.25 ppm)[3] | ||
| REL (Recommended) | TWA 1 mg/m3 (0.25 ppm)[three] | ||
| IDLH (Immediate danger) | 10 mg/thousandthree [iii] | ||
| Safety data sheet (SDS) | MSDS at J. T. Baker | ||
| Related compounds | |||
| Related acrid anhydrides | Succinic anhydride | ||
| Related compounds | Maleic acid | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |||
Maleic anhydride is an organic chemical compound with the formula C2Hii(CO)2O. Information technology is the acrid anhydride of maleic acid. Information technology is a colorless or white solid with an acrid smell. It is produced industrially on a large scale for applications in coatings and polymers.[5]
Production [edit]
Maleic anhydride is produced by vapor-phase oxidation of n-butane. The overall process converts the methyl groups to carboxylate and dehydrogenates the courage. The selectivity of the process reflects the robustness of maleic anhydride, with its conjugated double-bond arrangement. Traditionally maleic anhydride was produced past the oxidation of benzene or other effluvious compounds. As of 2006, only a few smaller plants continue to use benzene.
In both cases, benzene and butane are fed into a stream of hot air, and the mixture is passed through a goad bed at high temperature. The ratio of air to hydrocarbon is controlled to prevent the mixture from igniting. Vanadium pentoxide and molybdenum trioxide are the catalysts used for the benzene route, whereas vanadium phosphate is used for the butane route:[5]
- CivH10 + iii.5 Otwo → CfourH2O3 + iv H2O ∆H = −1236 kJ/mol
The main competing process entails full combustion of the butane, a conversion that is twice as exothermic equally the partial oxidation.
The traditional method using benzene became uneconomical due to the high and still rise benzene prices and by complying with the regulations of benzene emissions. In addition, in the production of maleic anhydride (4 C-atoms) a third of the original carbon atoms is lost every bit carbon dioxide when using benzene (half-dozen carbon atoms). The modern catalytic processes showtime from a 4-carbon molecule and merely attaches oxygen and removes h2o; the 4-C-base body of the molecule remains intact. Overall, the newer method is therefore more than fabric efficient.[vi]
Parallels be with the product of phthalic anhydride: While older methods use naphthalene, modern methods utilize o-xylene as feedstock.
Reactions [edit]
The chemistry of maleic anhydride is very rich, reflecting its ready availability and bifunctional reactivity. It hydrolyzes, producing maleic acid, cis-HOOC–CH=CH–COOH. With alcohols, the one-half-ester is generated, e.g., cis-HOOC–CH=CH–COOCH3.
Maleic anhydride is a archetype substrate for Diels-Alder reactions.[7] It was used for work in 1928, on the reaction between maleic anhydride and 1,3-butadiene, for which Otto Paul Hermann Diels and Kurt Alder were awarded the Nobel Prize in 1950. It is through this reaction that maleic anhydride converted to many pesticides and pharmaceuticals.
Michael reaction of maleic anhydride with agile methylene or methine compounds such equally malonate or acetoacetate esters in the presence of sodium acetate goad. These intermediates were subsequently used for the generation of the Krebs cycle intermediates aconitic and isocitric acids.[viii]
Maleic anhydride dimerizes in a photochemical reaction to form cyclobutane tetracarboxylic dianhydride (CBTA). This compound is used in the production of polyimides and as an alignment film for liquid crystal displays.[9]
It is too a ligand for depression-valent metal complexes, examples existence Pt(PPh3)ii(MA) and Fe(CO)iv(MA).
On account of its bike of 4 π electrons in an array of 5 atoms with p orbitals, maleic anhydride was long idea to exhibit antiaromaticity. Nevertheless, a thermochemical study concluded that just viii kJ/mol of destabilization energy tin can be ascribed to this effect, making information technology weakly antiaromatic at best.[10]
Uses [edit]
Maleic anhydride is used in many applications.[5]
Plastics & resins [edit]
Around 50% of world maleic anhydride output is used in the manufacture of unsaturated polyester resins (UPR). Chopped glass fibers are added to UPR to produce fiberglass reinforced plastics that are used in a broad range of applications such every bit pleasure boats, bathroom fixtures, automobiles, tanks and pipes.
Maleic anhydride is hydrogenated to i,iv-butanediol (BDO), used in the production of thermoplastic polyurethanes, elastane/Spandex fibers, polybutylene terephthalate (PBT) resins and many other products.
Curing agents [edit]
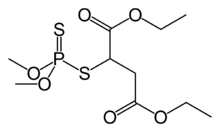
Malathion is a pop insecticide that is derived from maleic anhydride.

Diels-Alder reaction of maleic anhydride and butadiene and isoprene gives the respective tetrahydrophthalic anhydrides which can be hydrogenated to the corresponding hexahydrophthalic anhydrides. These species are used as curing agents in epoxy resins. Another market for maleic anhydride is lubricating oil additives, which are used in gasoline and diesel engine crankcase oils every bit dispersants and corrosion inhibitors. Changes in lubricant specifications and more than efficient engines have had a negative event on the demand for lubricating oil additives, giving flat growth prospects for maleic anhydride in this application.
Others [edit]
A number of smaller applications for maleic anhydride. The food industry uses malic acid which is derivative of maleic anhydride in artificial sweeteners and flavor enhancements. Personal care products consuming maleic anhydride include hair sprays, adhesives and floor polishes. Maleic anhydride is also a precursor to compounds used for h2o treatment detergents, insecticides and fungicides, pharmaceuticals, and other copolymers.
Major producers [edit]
| Company | Location | Capacity (KMT/Year) |
|---|---|---|
| Yongsan Chemicals, Inc. | Republic of korea | 38 |
| Bartek Ingredients Inc. | Canada | 28 |
| Sasol-Huntsman | Germany | 105 |
| DSM NV | The Netherlands | 100 |
| INEOS | Usa | l |
| Huntsman Corporation | Usa | 155 |
| Huntsman Performance Products | United states | 100 |
| Lanxess Corporation | Usa | 75 |
| Lonza Group AG | Switzerland | 100 |
| AOC Materials | Us | 55 |
| Mitsubishi Chemical Corporation | Nihon | 32 |
| Mitsui Chemicals, Inc | Japan | 33 |
| Mitsui Chemicals Polyurethanes, Inc. | Japan | 100 |
| Nippon Shokubai Co., Ltd | Nihon | 35 |
| NOF Corporation | Japan | 12 |
| Polynt SpA | Italian republic | 96 |
| Mysore Petro Chemicals Ltd. | India | 15 |
Source: Kirk & Othmer
Solid State Chemicals, Ltd. started product of solid maleic anhydride pastilles in the Us in 2014.
Globe Maleic Anhydride Capacity Past Region
Data in: kilotonnes per annum
| Region | 2002 | 2012 | 2015 (KMT/Year) |
|---|---|---|---|
| N America | 235 | 311 | 370 |
| South & Central America | 44 | 41 | 46 |
| Western Europe | 168 | 456 | 307 |
| Primal & Eastern Europe | 64 | 58 | 60 |
| Asia | 315 | 483 | 1864 |
| Africa | x | 10 | 14 |
| Total | 836 | 1359 | 2771 |
Source: Kirk & Othmer [ full citation needed ]
Packing and send [edit]
Liquid maleic anhydride is available in road tankers and/or tank-containers which are made of stainless steel, which are insulated and provided with heating systems to maintain the temperature of 65-75 °C. Tank cars must exist approved for the send of molten maleic anhydride.
Liquid/molten maleic anhydride is a dangerous cloth in accordance with RID/ADR.
Solid maleic anhydride pellets are transported by trucks. Packaging is generally in 25 kg polyethylene bags.
Effects on human health and the environment [edit]
This chemical compound poses relatively depression-risk environmental hazards, an of import feature for some applications. In humans, exposure to maleic anhydride may cause irritation to the respiratory tract, eyes, exposed mucosa, and pare. Maleic anhydride is besides a skin and respiratory sensitizer.[11]
Maleic anhydride is a depression take a chance profile chemical. Maleic anhydride quickly hydrolyzes to grade maleic acid in the presence of water and hence environmental exposures to maleic anhydride itself are unlikely. Maleic acid is biodegradable under aerobic conditions in sewage sludge as well as in soil and h2o.
Food starch for utilize in dark markets sold from a supplier in Tainan city, Taiwan, were found to contain maleic anhydride in Dec 2013. The supplier was investigated regarding the 300 tons of tainted starch; an earlier inspection in November had institute 32 tons.[12]
References [edit]
- ^ Merck Index, 11th Edition, 5586.
- ^ a b "Front Matter". Nomenclature of Organic Chemical science : IUPAC Recommendations and Preferred Names 2013 (Bluish Book). Cambridge: The Royal Society of Chemical science. 2014. p. 835. doi:x.1039/9781849733069-FP001. ISBN978-0-85404-182-4.
- ^ a b c d e f g NIOSH Pocket Guide to Chemical Hazards. "#0376". National Institute for Occupational Safety and Health (NIOSH).
- ^ "Maleic anhydride". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Wellness (NIOSH).
- ^ a b c Kurt Lohbeck; Herbert Haferkorn; Werner Fuhrmann; Norbert Fedtke. "Maleic and Fumaric Acids". Ullmann's Encyclopedia of Industrial Chemical science. Weinheim: Wiley-VCH. doi:10.1002/14356007.a16_053.
- ^ Bertram Philipp, Peter Stevens: Grundzüge der Industriellen Chemie, VCH Verlagsgesellschaft mbH, 1987, S. 179, ISBN 3-527-25991-0.
- ^ Samuel Danishefsky; Takeshi Kitahara & Paul F. Schuda (1983). Preparation and Diels-Alder Reaction of a Highly Nucleophilic Diene: trans-1-Methoxyl-3-Trimethylsiloxy-1,iii-Butadiene and 5β-Methoxycyclohexan-one-one-3β,4β-Dicarboxylic acid Andhydride. Org. Synth. Vol. 61. p. 147. doi:ten.1002/0471264180.os061.30. ISBN978-0471264224.
- ^ US 4146543 E.Gutierrez
- ^ Horie, T.; Sumino, M.; Tanaka, T.; Matsushita, Y.; Ichimura, T.; Yoshida, J. I. (2010). "Photodimerization of Maleic Anhydride in a Microreactor Without Clogging". Organic Procedure Research & Evolution. 14 (2): 100128104701019. doi:10.1021/op900306z.
- ^ Roux, María Victoria; Jiménez, Pilar; Martín-Luengo, Maria Ángeles; Dávalos, Juan Z.; Sunday, Zhiyuan; Hosmane, Ramachandra S.; Liebman, Joel F. (May 1997). "The Elusive Antiaromaticity of Maleimides and Maleic Anhydride: Enthalpies of Germination ofN-Methylmaleimide,N-Methylsuccinimide,N-Methylphthalimide, andN-Benzoyl-N-methylbenzamide". The Journal of Organic Chemistry. 62 (9): 2732–2737. doi:10.1021/jo9621985. ISSN 0022-3263. PMID 11671632.
- ^ "Substance Evaluation Written report: Maleic anhydride" (PDF). Surround Bureau Austria. Archived from the original (PDF) on 2014-10-18. Retrieved 2014-x-13 .
- ^ "Tainted starch found in Tainan yet again". Want China Times. 2013-12-xix. Archived from the original on 2013-12-19. Retrieved 2013-12-19 .
External links [edit]
- International Chemical Safety Card 0799
- NIOSH Pocket Guide to Chemic Hazards. "#0376". National Found for Occupational Condom and Health (NIOSH).
- Chronic toxicity summary
- Maleic anhydride at Occupational Rubber & Health Administration
Furan And Maleic Anhydride Reaction,
Source: https://en.wikipedia.org/wiki/Maleic_anhydride
Posted by: hobbsenee1983.blogspot.com







0 Response to "Furan And Maleic Anhydride Reaction"
Post a Comment