Cu No3 2 Chemical Name
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name Copper(Ii) nitrate | |||
| Other names Cupric nitrate | |||
| Identifiers | |||
| CAS Number |
| ||
| 3D model (JSmol) |
| ||
| ChEBI |
| ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.019.853 | ||
| PubChem CID |
| ||
| RTECS number |
| ||
| UNII |
| ||
| CompTox Dashboard (EPA) |
| ||
| InChI
| |||
| SMILES
| |||
| Properties | |||
| Chemical formula | Cu(NO3)2 | ||
| Tooth mass | 187.5558 g/mol (anhydrous) 241.threescore g/mol (trihydrate) 232.591 m/mol (hemipentahydrate) | ||
| Appearance | bluish crystals hygroscopic | ||
| Density | iii.05 thousand/cm3 (anhydrous) 2.32 g/cm3 (trihydrate) 2.07 g/cmiii (hexahydrate) | ||
| Melting point | 114 °C (237 °F; 387 K) (anhydrous, decomposes) 114.5 °C (trihydrate) 26.four °C (hexahydrate, decomposes) | ||
| Boiling point | 170 °C (338 °F; 443 Grand) (trihydrate, decomposes) | ||
| Solubility in water | trihydrate:[iii] 381 1000/100 mL (40 °C) 666 k/100 mL (80 °C) hexahydrate:[three] 243.7 g/100 mL (80 °C) | ||
| Solubility | hydrates very soluble in ethanol, ammonia, water; insoluble in ethyl acetate | ||
| Magnetic susceptibility (χ) | +1570.0·10−6 cm3/mol (~3HtwoO) | ||
| Structure | |||
| Crystal structure | orthorhombic (anhydrous) rhombohedral (hydrates) | ||
| Hazards | |||
| Occupational safety and wellness (OHS/OSH): | |||
| Principal hazards | Irritant, Oxidizer | ||
| NFPA 704 (burn down diamond) | 1 0 three OX | ||
| NIOSH (Usa health exposure limits): | |||
| PEL (Permissible) | TWA ane mg/grand3 (as Cu)[4] | ||
| REL (Recommended) | TWA i mg/1000three (as Cu)[four] | ||
| IDLH (Firsthand danger) | TWA 100 mg/m3 (as Cu)[4] | ||
| Safety information sheet (SDS) | Cu(NOiii)ii·3HiiO | ||
| Related compounds | |||
| Other anions | Copper(2) sulfate Copper(Two) chloride | ||
| Other cations | Argent nitrate Aureate(3) nitrate | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |||
Copper(Ii) nitrate describes any member of the family unit of inorganic compounds with the formula Cu(NO3)2(HiiO)x. The hydrates are blue solids. Anhydrous copper nitrate forms blue-dark-green crystals and sublimes in a vacuum at 150-200 °C.[5] Common hydrates are the hemipentahydrate and trihydrate.
Synthesis and reactions [edit]
Hydrated copper(II) nitrate [edit]
Hydrated copper nitrate is prepared past treating copper metallic or its oxide with nitric acid:[6]
- Cu + ivHNO3 → Cu(NO3)two . + 2H2O + 2NOii
The same salts can be prepared treating copper metallic with an aqueous solution of silver nitrate. That reaction illustrates the ability of copper metal to reduce silver ions.
In aqueous solution, the hydrates exist every bit the aquo circuitous [Cu(H2O)6]2+. Such complexes are highly labile owing to the d9 electronic configuration of copper(II).
Attempted dehydration of whatever of the hydrated copper(II) nitrates by heating affords the oxides, not Cu(NOiii)2. At lxxx °C, the hydrates convert to "bones copper nitrate" (Cu2(NO3)(OH)3), which converts to CuO at 180 °C.[6] Exploiting this reactivity, copper nitrate can be used to generate nitric acid past heating it until decomposition and passing the fumes direct into water. This method is similar to the last pace in the Ostwald process. The equations are every bit follows:
- 2 Cu(NO3)2 → 2 CuO + 4 NOii + O2
- 3NO2 + HiiO → 2HNO3 + NO
Treatment of copper(Two) nitrate solutions with triphenylphosphine, triphenylarsine, and triphenylstibine gives the corresponding copper(I) complexes [Cu(E(CviH5)3)three]NO3 (E = P, Equally, Sb). The group V ligand is oxidized to the oxide.[7]
Anhydrous copper(II) nitrate [edit]
Two polymorphs of anhydrous Cu(NO3)2 are known. The and then-called β-form is a covalent molecular circuitous, as evidenced by its tendency to sublime. It is one of the few anhydrous transition metallic nitrates.[8] Information technology cannot be prepared past reactions containing or producing water. Instead, anhydrous Cu(NO3)ii forms when copper metal is treated with dinitrogen tetroxide:
- Cu + 2 Due north2O4 → Cu(NOiii)2 + 2 NO
The α-grade of anhydrous copper(II) nitrate is a coordination polymer.[1]
Structure [edit]
Anhydrous copper(2) nitrate [edit]
-
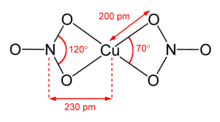
Structure of anhydrous copper(2) nitrate.
Anhydrous copper(2) nitrate has been crystallized in ii solvate-free polymorphs.[i] [2] α- and β-Cu(NOiii)ii are fully 3D coordination polymer networks. The alpha form has only one Cu surround, with [4+1] coordination, just the beta form has two different copper centers, one with [4+1] and 1 that is foursquare planar. The nitromethane solvate also features "[iv+ 1] coordination", with 4 short Cu-O bonds of approximately 200 pm and one longer bail at 240 pm.[ix] They are coordination polymers, with space bondage of copper(II) centers and nitrate groups. In the gas phase, copper(Ii) nitrate features ii bidentate nitrate ligands (see image at upper correct).[10] Thus, evaporation of the solid entails "bully" to requite the copper(II) nitrate molecule.
Hydrated copper(2) nitrate [edit]
Five hydrates have been reported: the monohydrate (Cu(NOthree)2·H2O),[2] the sesquihydrate (Cu(NO3)ii·ane.5H2O),[xi] the hemipentahydrate (Cu(NO3)2·ii.5H2O),[12] a trihydrate (Cu(NOiii)ii·3HtwoO),[xiii] and a hexahydrate ([Cu(HtwoO)half dozen](NO3)2).[14] The hexahydrate is interesting because the Cu-O distances are all equal, non revealing the usual effect of Jahn-Teller distortion that is otherwise feature of octahedral Cu(Two) complexes. This non-issue is attributed to the strong hydrogen bonding that limits the elasticity of the Cu-O bonds.
Applications [edit]
Copper(Ii) nitrate finds a variety of applications, the main 1 being its conversion to copper(II) oxide, which is used equally catalyst for a diversity of processes in organic chemical science. Its solutions are used in textiles and polishing agents for other metals. Copper nitrates are institute in some pyrotechnics.[half dozen] It is often used in school laboratories to demonstrate chemical voltaic cell reactions. It is a component in some ceramic glazes and metal patinas.
Organic synthesis [edit]
Copper nitrate, in combination with acerb anhydride, is an constructive reagent for nitration of aromatic compounds, known as the Menke nitration.[15] Hydrated copper nitrate adsorbed onto clay affords a reagent chosen "Claycop". The resulting blue-colored dirt is used as a slurry, for example for the oxidation of thiols to disulfides. Claycop is also used to convert dithioacetals to carbonyls.[16] A related reagent based on montmorillonite has proven useful for the nitration of aromatic compounds.[17]
Naturally occurring copper nitrates [edit]
No mineral of the ideal Cu(NOiii)2 formula, or the hydrate, are known. Likasite, Cu3(NO3)(OH)five .2H2O and buttgenbachite, Cu19(NO3)2(OH)32Cl4·2HiiO are related minerals.[18] [nineteen]
Natural basic copper nitrates include the rare minerals gerhardtite and rouaite, both beingness polymorphs of Cu2(NO3)(OH)iii substance.[20] [21] [22] A much more than complex, basic, hydrated and chloride-bearing natural salt is buttgenbachite.[19] [22]
References [edit]
- ^ a b c Wallwork, S. C.; Addison, W. E. (1965). "526. The crystal structures of anhydrous nitrates and their complexes. Part I. The α form of copper(Ii) nitrate". J. Chem. Soc. 1965: 2925–2933. doi:ten.1039/JR9650002925.
- ^ a b c Troyanov, S. I.; Morozov, I. Five.; Znamenkov, K. O.; Yu; Korenev, Chiliad. (1995). "Synthesis and X-Ray Structure of New Copper(II) Nitrates: Cu(NO3)two·HtwoO and ?-modification of Cu(NO3)two". Z. Anorg. Allg. Chem. 621 (7): 1261–1265. doi:10.1002/zaac.19956210727.
- ^ a b Perrys' Chem Eng Handbook, seventh Ed
- ^ a b c NIOSH Pocket Guide to Chemic Hazards. "#0150". National Institute for Occupational Safety and Health (NIOSH).
- ^ Pass and Sutcliffe (1968). Practical Inorganic Chemistry. London: Chapman and Hall.
- ^ a b c H.Wayne Richardson "Copper Compounds" Ullmann's Encyclopedia of Industrial Chemistry 2005, Wiley-VCH, Weinheim. doi:ten.1002/14356007.a07_567.
- ^ Gysling, Henry J. (1979). "Coordination Complexes of Copper(I) Nitrate". Inorganic Syntheses. Inorganic Syntheses. Vol. 19. pp. 92–97. doi:10.1002/9780470132500.ch19. ISBN9780470132500.
- ^ Addison, C. C.; Logan, N.; Wallwork, S. C.; Garner, C. D. (1971). "Structural Aspects of Co-ordinated Nitrate Groups". Quarterly Reviews, Chemical Society. 25 (2): 289. doi:ten.1039/qr9712500289.
- ^ Duffin, B.; Wallwork, S. C. (1966). "The crystal construction of anhydrous nitrates and their complexes. II. The 1:i copper(Ii) nitrate-nitromethane complex". Acta Crystallographica. 20 (ii): 210–213. doi:10.1107/S0365110X66000434.
- ^ LaVilla, R. E.; Bauer, S. H. (1963). "The Construction of Gaseous Copper(Ii) Nitrate equally Determined by Electron Diffraction". J. Am. Chem. Soc. 85 (22): 3597–3600. doi:10.1021/ja00905a015.
- ^ Dornberger-Schiff, Yard.; Leciejewicz, J. (1958). "Zur Struktur des Kupfernitrates Cu(NOiii)ii .1.5HiiO". Acta Crystallogr. 11 (11): 825–826. doi:ten.1107/S0365110X58002322.
- ^ Morosin, B. (1970). "The crystal structure of Cu(NOthree)2.ii.5H2O". Acta Crystallogr. B26 (9): 1203–1208. doi:10.1107/S0567740870003898.
- ^ J. Garaj, Sbornik Prac. Chem.-Technol. Fak. Svst., Cskosl. 1966, pp. 35–39.
- ^ Zibaseresht, R.; Hartshorn, R. M. (2006). "Hexaaquacopper(II) dinitrate: absence of Jahn-Teller distortion". Acta Crystallogr. E62: i19–i22. doi:10.1107/S1600536805041851.
- ^ Menke J.B. (1925). "Nitration with nitrates". Recueil des Travaux Chimiques des Pays-Bas. 44: 141. doi:10.1002/recl.19250440209.
- ^ Balogh, 1000. "Copper(2) Nitrate–K10 Bentonite Clay" in Encyclopedia of Reagents for Organic Synthesis (Ed: L. Paquette) 2004, J. Wiley & Sons, New York. doi:x.1002/047084289X.
- ^ Collet, Christine (1990). "Clays Direct Aromatic Nitration". Angewandte Chemie International Edition in English. 29 (five): 535–536. doi:10.1002/anie.199005351.
- ^ "Likasite". www.mindat.org.
- ^ a b "Buttgenbachite". world wide web.mindat.org.
- ^ "Gerhardtite". www.mindat.org.
- ^ "Rouaite". www.mindat.org.
- ^ a b International Mineralogical Association (21 March 2011). "Listing of Minerals". www.ima-mineralogy.org.
External links [edit]
- National Pollutant Inventory – Copper and compounds fact canvas
- ICSC Copper and compounds fact canvas
Cu No3 2 Chemical Name,
Source: https://en.wikipedia.org/wiki/Copper%28II%29_nitrate
Posted by: hobbsenee1983.blogspot.com





0 Response to "Cu No3 2 Chemical Name"
Post a Comment